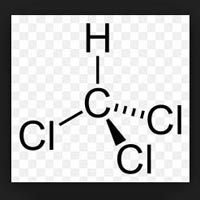
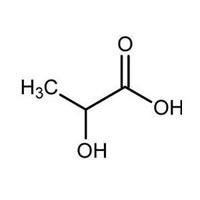
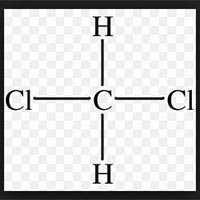
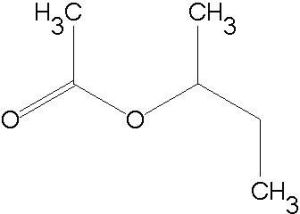
| Business Type | Manufacturer, Exporter, Supplier |
Acetonitrile (ACN) is the chemical compound with formula CH3CN. This colourless liquid is the simplest organic nitrile. It is produced mainly as a by product of acrylonitrile manufacture. It is widely used as a polar aprotic solvent in synthetic chemistry, and as a medium-polarity solvent in HPLC.
Acetonitrile is mainly used as a solvent that is miscible with water and has a convenient liquid range. With a dipole moment of 3.84 D, acetonitrile dissolves a wide range of ionic and nonpolar compounds and with its low reactivity, it does so with minimal complications.
It is widely used in battery applications because of its relatively high dielectric constant and ability to dissolve electrolytes. For similar reasons it is a popular solvent in cyclic voltammetry. Its low viscosity and low chemical reactivity make it a popular choice for liquid chromatography.
Packing: In 150 kg Iron drums