| Business Type | Exporter, Supplier |
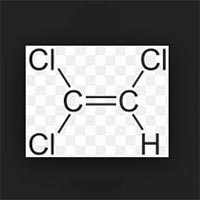
Xylene (C8H10), any of three isomeric dimethylbenzenes which have the same chemical formula, C6H4(CH3)2, but different molecular structure. All three are obtained from coal-tar distillate and petroleum as a mixture containing 50–60 percent by volume of m-xylene and 20–25 percent of each of the other isomers. The commercial xylene mixture is a colourless, nonviscous, flammable, toxic liquid that is insoluble in water but miscible with many organic liquids.
Uses :
Xylene is commonly used as a solvent for lacquers and rubber cements. It is also used as solvents, as components of aviation fuel, and as raw materials for the manufacture of dyes, fibres, and films. Meta- and para-xylene undergo nitration and reduction to give xylidines and used in making dyes. The meta isomer also is converted to trinitro-t-butyl-m-xylene, or xylene musk, a component of perfumes. Oxidation of the xylenes gives monocarboxylic acids (toluic acids), and then dicarboxylic acids (phthalic acids).